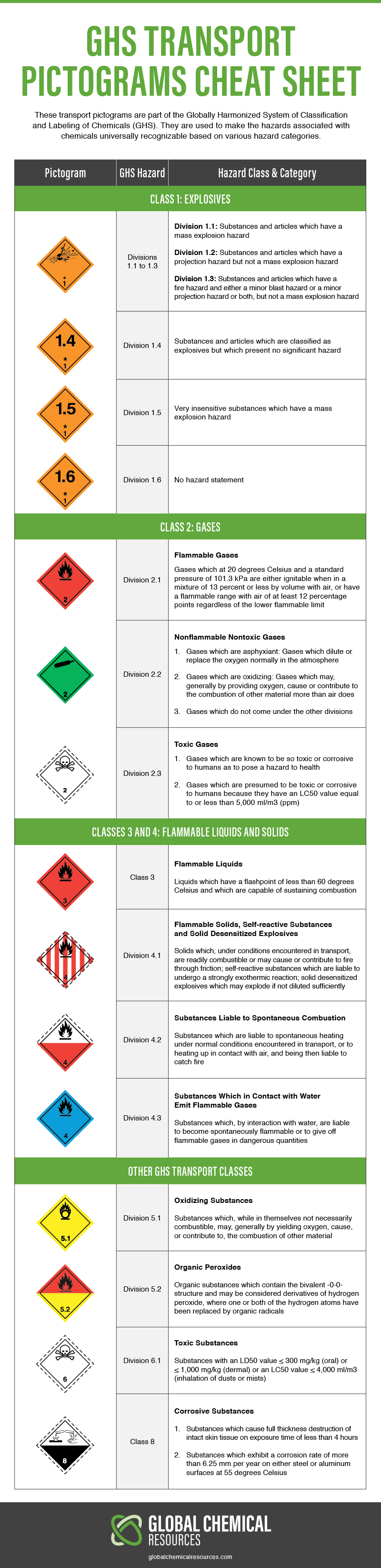
Strong Oxidizer Haz Mat
In one sense an oxidizing agent is a chemical species that undergoes a chemical reaction in which it gains one or more electrons.
Strong oxidizer haz mat. 09sept2016 2 increases often leading to a fire or explosion. This chemical material is a mix between strong oxidizing agents perchlorate and a good fuel ammonium. Chlorine is a yellowish green gas compound that has stinging smell and poisonous when there is direct contact. An oxidizing agent often referred to as an oxidizer or an oxidant is a chemical species that tends to oxidize other substances i e.
In chemistry an oxidising agent oxidant oxidizer is a substance that has the ability to oxidize other substances in other words to accept their electrons common oxidizing agents are oxygen hydrogen peroxide and the halogens. Ammonium perchlorate is a chemical compound that is used as an exploding materials and fireworks as a rocket oxidizing agent glue etc. This means that oxygen combines chemically with the other material in a way that increases the chance of a fire or explosion. In the case of oxidizing acids the hazard is very high and the ehs sop corrosive chemical hazards risk minimization should be followed as well.
This class of chemicals includes peroxides chlorates perchlorates nitrates and permanganates. Seek more specific information on the reactivity of particular chemicals when a procedure requires mixing a strong oxidizer with an organic chemical. Use oxidizers in a chemical fume hood. Oxidizing materials are liquids or solids that readily give off oxygen or other oxidizing substances such as bromine chlorine or fluorine.
Strong oxidizers are capable of forming explosive mixtures. Common examples of oxidizing agents include halogens such as chlorine and fluorine oxygen and hydrogen peroxide h 2 o 2. Common chemical oxidizing agents chlorine is a green color member of viia group that can not be found freely in nature but in a combined form such as nacl strong electrolyte or in mineral formations such as sylvite and carnallite. They also include materials that react chemically to oxidize combustible burnable materials.
Explosive and strong oxidizer. Chemical storage and transportation in this chp for a complete list of requirements. Oxidizers and oxidizing gases should have a boundary separation of 8 10 m or a 1 hour fire wall from flammable or reactive materials. Since the purpose of oxidizers is to oxidize tissues such as lung skin and eyes are at risk.
Cause an increase in the oxidation state of the substance by making it lose electrons. Prior to working with this chemical personnel should be trained on its. If there is a risk of explosion or violent reaction it is absolutely necessary to use the hood sash as a protective. Specially designed corrosion resistant cabinets should be used for the storage of corrosive materials.
The hazards to tissues from other oxidizers will vary depending on the oxidizer and its concentration. Store separately from all other flammable materials. Both explains how ammonium perchlorate can be used as a rocket fuel.